Abstract
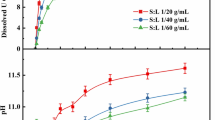
The dissolution rate of unirradiated uranium dioxide was studied in batch experiments as a function of hydrogen peroxide concentration (from 10−5 to 10−3 mol dm−3). Unirradiated UO2(s) was used in order to differentiate surface chemical processes from radiolytic effects. Dissolution rates were determined from both uranium release and hydrogen peroxide consumption. Results showed that H2O2consumption rate was higher than UO2 dissolution rate. This observation may indicate that the overall UO2 oxidative dissolution process would be controlled by the dissolution of the oxidized solid surface. The calculated hydrogen peroxide reaction order was 1 in the H2O2 concentration range from 10−5 to 10−4 mol dm−3, while at higher concentrations no clear dependence was observed.
Similar content being viewed by others
References
T. Eriksen, U.B. Ekulnd, L. Werme and J. Bruno, J. of Nucl. Mat. 227, 76–82 (1995).
B. Grambow, A. Loida, P. Dressier, H. Geckeis, J. Gago, I. Casas, J. de Pablo, J. Giménez and M.E. Torrero, Chemical reaction of fabricated and high burn-up spent UO2 fuel with saline brines, European Comission, Final Report EUR 17111 (1997) pp 166
W.J Gray and S.A. Steward, J. Nucl. Mater. 190, 46 (1992).
W.J. Gray and C.N. Wilson, (1995) Spent fuel dissolution studies: FY 1991 to 1994. Report PNL-10540 (USA).
M. E. Torrero, E. Baraj, J. de Pablo, J. Giménez and I. Casas, Int. J. Chem. Kinetic 29, 261–267 (1997)
D. W. Shoesmith and S. Sunder, J. of Nucl. Mat. 190, 20–35 (1992).
J. de Pablo, I. Casas, J. Gimenez, M. Molera, M. Rovira, L. Duro and J. Bruno, Geochim. et Cosmochim. Acta 63, 3097–3103 (1999)
J.B. Hiskey, Trans. Inst. Nin. Metall. Sect. C, 88, C145 (1979).
J. Giménez, E. Baraj, M.E. Torrero, I. Casas and J. de Pablo, J. Nucl. Mater. 238, 64–69 (1996).
P. Diaz-Arocas, J. Quiñones, C. Maffiotte, J. Serrano, J. Garcia, J.R. Almazán and J. Esteban in Scientific Basis for Nuclear Waste Management XVIII, edited by T. Murakami and R.C. Ewing. (Mat. Res. Soc. Symp. Ser. 353, Pittsburg, PA 1995) pp. 641–646.
J.C. Robbins, Can. Inst. Min. Metall. Bull. 71, 61 (1978).
D. Price, P. J. Worsfold and R. F. C. Montoura, Anal. Chim. Acta 298, 121–128 (1994)
D.W. Shoesmith, S. Sunder, M.G. Mailey and G.J. Wallace, Corrosion Science 29, 1115 (1989).
I. Grenthe, J. Fuger, R.J.M. Konings, R.J. Lemire, A.B. Muller, C. Nguyen-Trung and H. Wanner: Chemical Thermodynamics of Uranium (eds. H. Wanner and I. Forest). Elsevier Sci. Publishers, Amsterdam 1992.
J. Bruno, I. Casas, E. Cera, J. de Pablo, J. Giménez and M.E. Torrero in Scientific Basis for Nuclear Waste Management XVIII, edited by T. Murakami and R.C. Ewing. (Mat. Res. Soc. Symp. Ser. 353, Pittsburg, PA 1995) pp. 601–608.
S. Sunder, D.W. Shoesmith, R.J. Lemire, M.G. Bailey and G.J. Wallace, Corros. Sci. 32, 373–386 (1991).
J. Bruno, E. Cera, L. Duro, J. Pon, J. de Pablo and T. Eriksen, Development of a kinetic model for the dissolution of the UO2 spent nuclear fuel, SKB TR-98-22, pp 49 (1998)
I. A. Salem, R. I. Elhag and K.M.S. Khalil, Transition Metal Chemistry 25, 260–264 (2000)
R. Andreozzi, V. Caprio, A. Insola and R. Marotta, Catalysis Today 53, 51–59 (1999)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
De Pablo, J., Casas, I., Clarens, F. et al. The Effect of Hydrogen Peroxide Concentration on the Oxidative Dissolution of Unirradiated Uranium Dioxide. MRS Online Proceedings Library 663, 409 (2000). https://doi.org/10.1557/PROC-663-409
Published:
DOI: https://doi.org/10.1557/PROC-663-409