Article contents
Synthesis of DNA-encapsulated silica elaborated by sol–gel routes
Published online by Cambridge University Press: 28 September 2012
Abstract
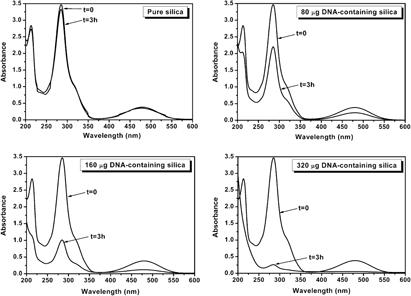
The highly specific functions of DNA can be used for designing novel functional materials. However, aqueous solubility and biochemical instability of DNA impede its direct utilization as a functional component. Herein, preparation of a hybrid material encapsulating the DNA molecules (double-stranded salmon sperm, 50–5000 base pairs) in robust host—sol–gel-derived silica—has been described. The encapsulation was carried out in two steps: hydrolysis of an acidic tetraethylorthosilicate [Si(OC2H5)4] sol and was followed by condensation near physiological pH upon addition of alkaline DNA-containing solutions. The gelation behavior and structural properties of the DNA–silica hybrids were investigated by 29Si nuclear magnetic resonance and by nitrogen adsorption. The selective adsorption of a DNA-interactive reagent molecule (ethidium bromide) in their diluted aqueous solutions on DNA–silica hybrids confirmed that the DNA molecules remained entrapped within the silica host without any deterioration. A DNA encapsulation mechanism correlating the silica microstructure and DNA holding efficiency has been proposed.
Keywords
- Type
- Articles
- Information
- Journal of Materials Research , Volume 28 , Issue 2: Focus Section: Silicon-based Nanoparticles for Biosensing and Biomedical Applications , 28 January 2013 , pp. 175 - 184
- Copyright
- Copyright © Materials Research Society 2012
References
REFERENCES
- 8
- Cited by




